Advantages of optical stimulation
Optogenetics enables the expression of light-sensitive channels for light-induced control of cellular function. Optogenetics is a powerful and versatile research tool. However, in vitro applications have been limited by low throughput light stimulation devices with rudimentary controls. The Lumos, introduced in 2016, provides flexible, powerful light delivery for in vitro applications at a multiwell high throughput scale. The Lumos allows for independent and simultaneous activation of 192 LEDs across 48 wells with user-defined control over wavelength, intensity, and microsecond duration.

Lumos MEA 48
Axion BioSystems’ Lumos MEA 48 plates combine high quality MEA results with highly optimized optical performance. The custom-formulated plate walls provide high reflectance to maximize light delivery to your culture and minimize well-to-well crosstalk. The custom-molded lid mates to the Lumos array and contains integrated optical lenses. Finally, the transparent bottom allows for cell visualization and assay multiplexing with fluorescence, luminescence, and other reporter-based assays.
The Lumos Advantage
- High throughput, multiwell format.
- Customized plate material and lid optics for optimal light delivery and even light dispersion.
- Transparent bottom for culture monitoring with light microscopy and assay multiplexing with fluorescence, luminescence, or other reporters.
- Independent control of light wavelength, intensity, and duration for each of 48 wells.
Improve the quality and consistency of your assay
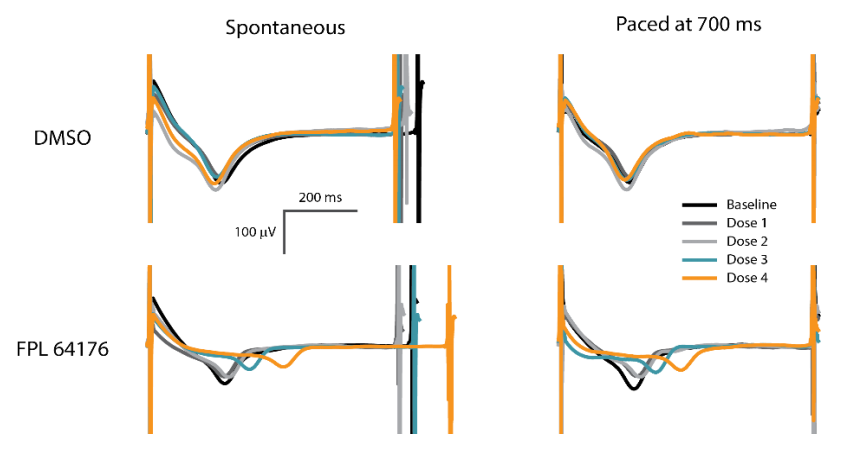
Addition of excitatory light-sensitive opsins, such as channelrhodopsin (ChR2), allows for controlled pacing of cardiomyocyte beating or neural network bursting. For cardiomyocytes, pacing the beat rate allows the user to increase physiological relevance, reduce well-to-well variability, and explore use-dependent drug effects.
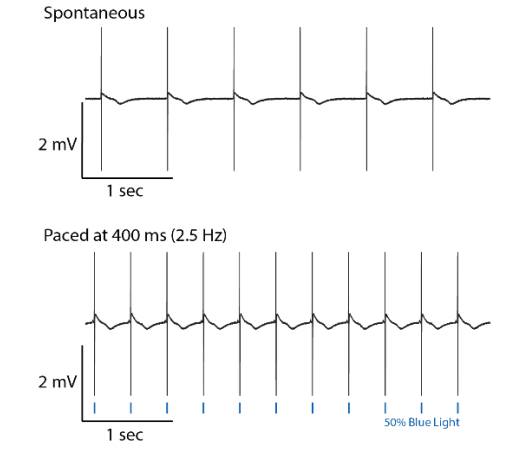
Discover more from your assay by controlling activity
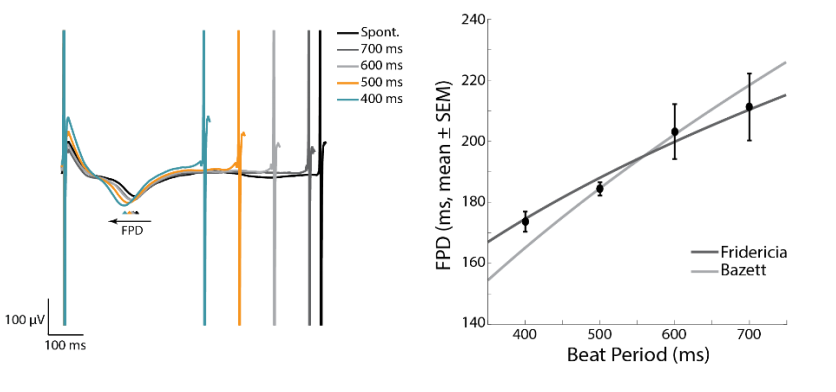
Many compounds simultaneously affect the beat rate and FPD of hiPSC-derived CMs. Optogenetic pacing enables the user to isolate the compound effects on repolarization alone by controlling the beat rate. Thus, pacing with the Lumos MEA plate significantly increases the information content of your assay.