Abstract
Cell signaling via G-protein coupled receptors (GPCRs), and the downstream cellular processes, cause changes to cell attachment and morphology detectable through impedance measurements. Here, using the Maestro Z platform, we studied the dynamic response of Calu-3 cells to Isoproterenol, Histamine, and Cytochalasin D, revealing important information on the kinetics and mechanism of action for each treatment. Furthermore, we characterized the downstream effect of Isoproterenol, and more generally cAMP activation, on the cystic fibrosis transmembrane conductance regulator (CFTR) present in the Calu-3 immortalized cell line.
Introduction
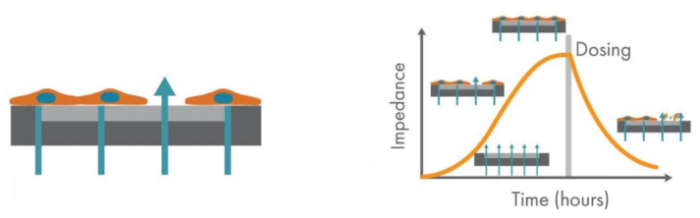
Cell signaling pathways communicate messages from outside the cell. When extracellular signal molecules bind receptors on the cell surface, they initiate signaling events inside the cell that determine cell behavior. G-protein-coupled receptors (GPCRs) are the largest class of transmembrane receptors. GPCR binding results in conformational changes and downstream responses that can be measured by impedance. These small changes often occur rapidly, but can last minutes to hours with significant physiological consequences. The Maestro Z impedance assay allows you to continuously measure the dynamics of cell signaling over minutes to hours without disturbing the cell biology (Figure 1).
Materials and Methods
Materials
Cells and reagents
Calu-3 (HTB-55) cells were obtained from ATCC (Manassas, VA). Media was prepared using MEM base media (Gibco, Cat. 11095072), 10% FBS (Gibco, Cat. 16000044), and 1% penicillin/streptomycin (Gibco, Cat. 15140122).
CytoView-Z Plate
A CytoView-Z 96-well plate (Axion BioSystems) was used for these experiments (Figure 2). The plate is composed of a polyethylene terephthalate (PET) surface with a gold recording electrode embedded in the culture surface of each well. Humidity reservoirs on the plate were filled with sterile deionized water to maintain humidity.
Maestro Z Impedance-based assay platform
The Maestro Z platform (Axion BioSystems) uses impedance measurements (ohms, Ω) to quantify the presence of cells on the electrode. To measure impedance, small electrical signals are delivered to the electrodes. Cell attachment, spreading, and cell-cell connections block these electrical signals and are detected as an increase in impedance. Impedance is also sensitive to subtle changes in cell conformation, such as those caused by receptor-mediated signaling or cell morphology. Since impedance is non-invasive and label-free, impedance assays can be used to quantify dynamic cellular responses over minutes, hours, and days. In addition, the Maestro Z platform has built-in environmental control, finely controlling temperature and CO2 throughout the experiment.
Methods
Cell plating
CytoView-Z plates were coated with 100 µL of fibronectin solution (1µg/mL) per well. Surface-coated impedance plates were incubated at 37°C and 5% CO2 for at least 1 hour. After incubation, excess surface coating was aspirated from each well. Then, 100 µL of complete medium was added per well and docked on the Maestro to record a media reference.
Cell Culture
Calu-3 cells were thawed and cultured according to the supplier recommendations, passaging as needed. Cells were lifted from flasks and dissociated via trypsinization. Cell density and viability was determined using a hemocytometer. The cell suspension was transferred to a 15 mL conical tube and centrifuged at 1,000 RPM for 5 minutes. The supernatant was aspirated, being careful to not disturb the cell pellet. Calu-3 cells were resuspended and diluted in media to a working concentration of 500,000 cells/ml.
Results
Isoproterenol Initiates Cell Signaling in Calu-3 Model
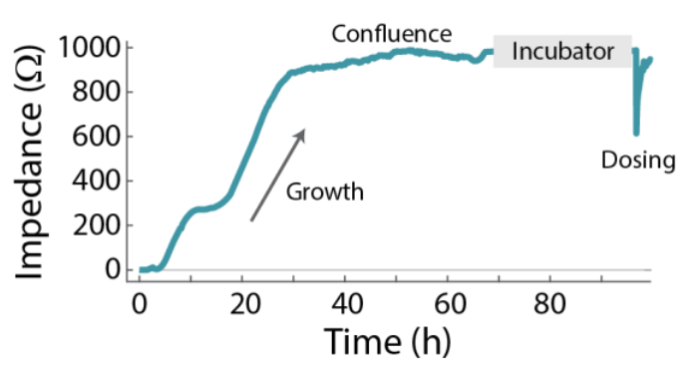
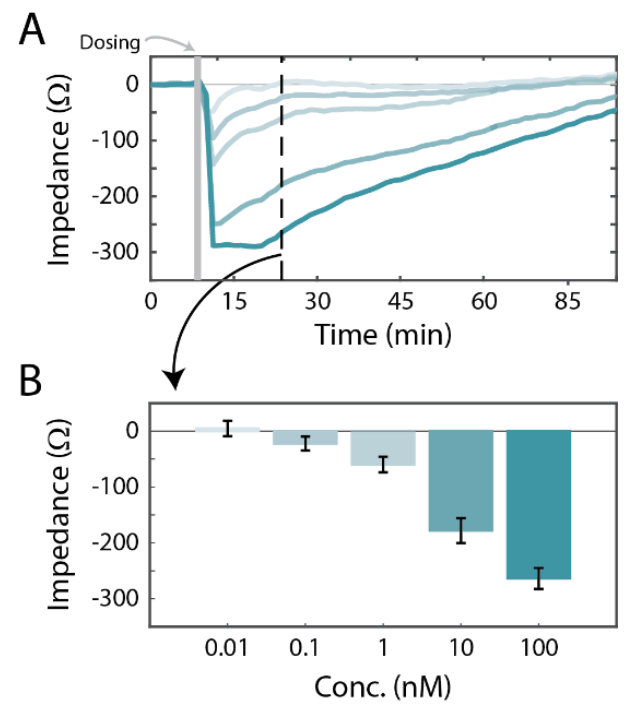
The Calu-3 cells were cultured on the Maestro Z platform until confluence, then treated with a concentration sweep of isoproterenol, a beta-adrenergic receptor agonist. Whereas the initial cell growth occurred on the time scale of days (Figure 2), the response to isoproterenol caused a significant change in impedance within a few minutes (Figure 3). The isoproterenol response was measured as a change relative to the impedance of the confluent monolayer. Framed in this way, the isoproterenol caused a dose-dependent decrease immediately following the live-dosing on the Maestro Z platform, as shown in Figure 3A. The dose response is illustrated in Figure 3B, at 20 minutes post-dose. The isoproterenol response subsequently decayed over time.
Dynamic Cell Response Varies with Mechanism
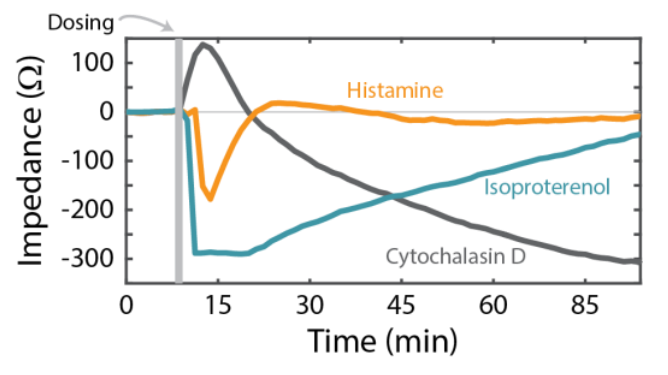
The Calu-3 cells were also treated with Histamine and Cytochalasin-D. Histamine, like Isoproterenol, is a GPCR agonist, but displayed a smaller, more transient response (orange, Figure 4). According to previous studies [Shen et al., 1994], the transient histamine response is consistent with an increase in intracellular Ca2+ activating barrier permeability (e.g., reducing the impedance). The sustained response to isoproterenol instead acts via cAMP signaling to stimulate ion transport and reduce impedance. By comparison, Cytochalasin-D is not a GPCR agonist. Rather, it inhibits actin polymerization and disrupts the cytoskeleton, causing a decrease in impedance (gray, Figure 4) over a longer timescale than histamine or isoproterenol.
Isoproterenol Activates Downstream Cellular Effects in Calu-3 Model
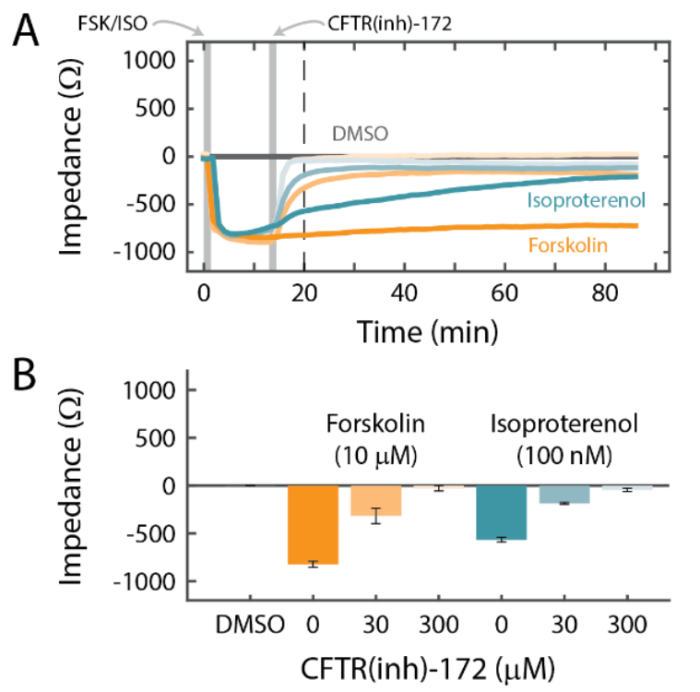
Calu-3 is one the few immortalized cell lines that produce tight junctions and express functional cystic fibrosis transmembrane conductance regulator (CFTR), which, upon beta-adrenergic stimulation, allows ion transport across the cell membrane [Shen et al., 1994]. In Figure 5, a significant decrease in impedance was detected following isoproterenol addition, consistent with activation of the CFTR. To further characterize the downstream cellular effects of isoproterenol, the Calu-3 cells were treated with a variety of pharmacological manipulations and monitored using the Maestro Z platform.
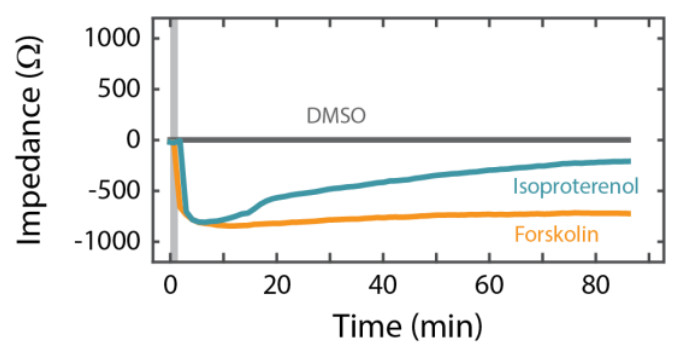
Addition of Forskolin, which directly activates cyclic AMP (cAMP), produced a similar response to Isoproterenol (Figure 6). Notably, however, the Forskolin response did not decrease over time like the Isoproterenol response, suggesting β2-adrenergic receptor desensitization or internalization. Activation via cAMP is consistent with CFTR function [Shen et al, 1994; Banga et al., 2014]. Further, the responses to Isoproterenol and Forksolin were eliminated in a dose-dependent manner by CFTR(inh)-172, a selective CFTR blocker (Figure 5). These results support that adrenergic stimulation of Calu-3 cells acts through a GPCR-mediated increase in cAMP to open pathways for ionic transport via the functional CFTR.
Conclusion
The Maestro Z platform was used to characterize the response of the Calu-3 cell line to perturbations causing rapid changes to cell morphology, using GPCR agonists. The degree and dynamics of the cellular response varied with the distinct mechanisms of action. The adrenergic response of the Calu-3 cells, and its association with functional CFTR expression, was further characterized and establishes the Maestro Z platform and Calu-3 cells as an in vitro model for studying barrier integrity.
References
1. Shen, Finkbeiner, Wine, Mrsny, Widdicombe, 1994 Am J Physiol Lung Cell Mol Physiol (5) p. L493-L501
2. Banga, Flaig, Lewis, Winfree, Blazer-Yost, 2014 Am J Physiol Lung Cell Mol Physiol. (10) p. L937-L946.
Authors
Daniel Millard, PhD
Anthony Nicolini, MSc
Colin Arrowood, MSc
Denise Sullivan, MSc
Heather Hayes, PhD
Axion BioSystems, Atlanta, GA





